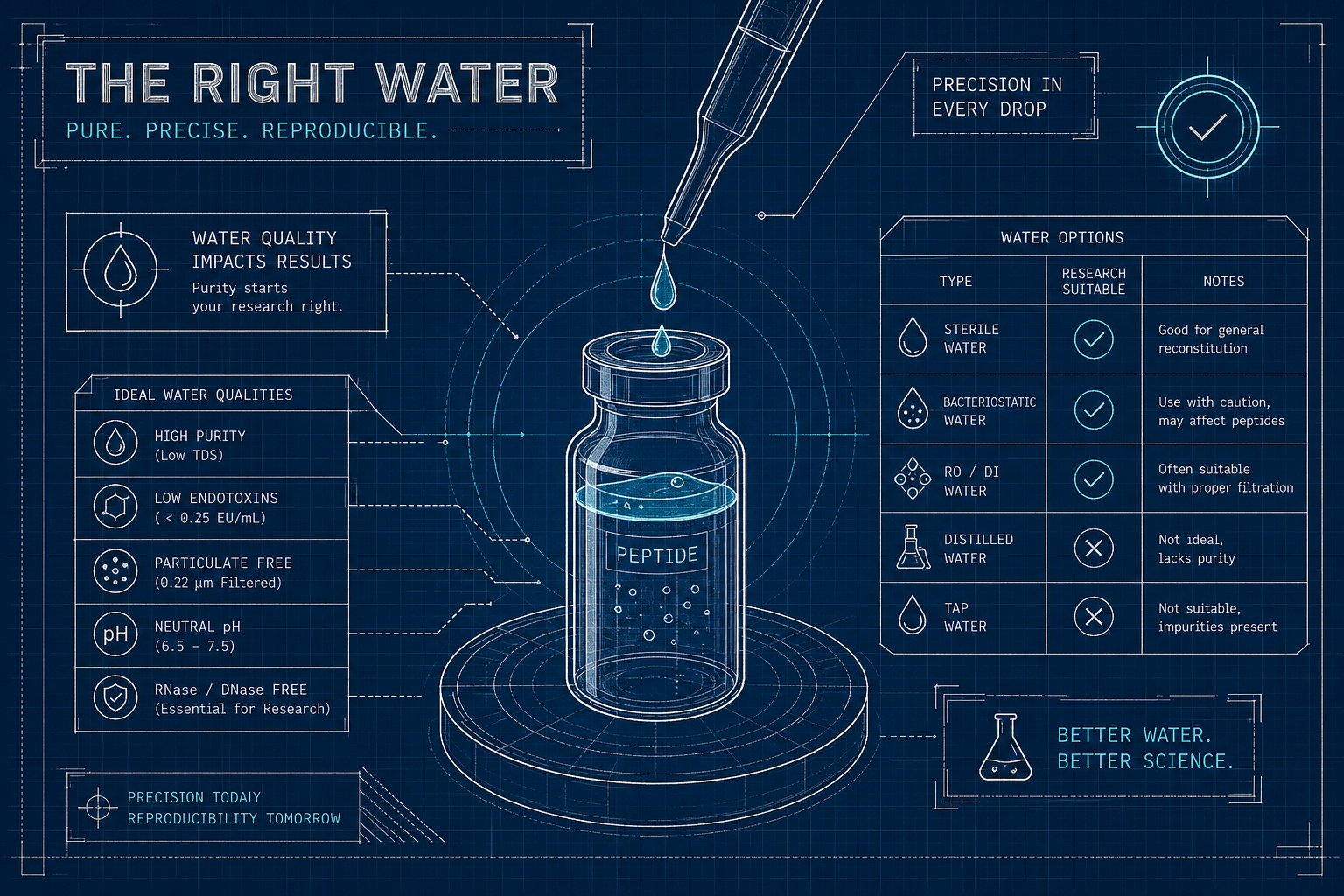
Use bacteriostatic water for multi-dose protocols — it contains 0.9% benzyl alcohol, which prevents bacterial growth between draws and gives you about 28 days of refrigerated stability. Use sterile water for injection for single-use experiments. Use PBS when the assay specifically requires physiological pH near 7.4. Match those defaults against the peptide's amino acid composition before you reconstitute.
The wrong solvent silently undermines everything that came before it. Researchers spend weeks vetting purity scores, cross-referencing COAs, and hunting for independent HPLC data, then grab whatever fluid is nearby when it is time to reconstitute. That mismatch can degrade the peptide before the first draw, introduce contamination that no freezer temperature will fix, or push a sensitive sequence out of solution entirely. Solvent selection deserves the same rigor as vendor selection — and a vendor's reconstitution guidance is itself a useful proxy for the quality of what is in the vial. We cover the broader vendor due-diligence pattern in our vendor red flags checklist.
This guide covers what is actually in each solvent, when to pick which one, how to match solvent to amino-acid composition, and what to do at storage time so the work survives the protocol.
What is in each solvent
The three solvents you will choose between are not interchangeable. Their chemistry differs in ways that matter for stability, contamination, and assay outcomes.
Bacteriostatic water (BAC water)
Sterile water containing 0.9% benzyl alcohol (9 mg/mL) as a preservative, per USP pharmaceutical specifications. The benzyl alcohol does not sterilize existing contamination — it inhibits new bacterial growth each time you pierce the septum. pH is mildly acidic, around 5.7, with an acceptable USP range of 4.5 to 7.0. Some peptide-specific BAC water formulations use 0.2% benzyl alcohol instead, to reduce potential preservative-peptide interaction. That detail is worth confirming when sourcing the solvent itself.
Sterile water for injection (SWFI)
Purified, non-pyrogenic water with no additives. Sterile when sealed; once opened, there is nothing preventing bacterial growth if the solution sits or is reused. SWFI is the right choice for single-use applications and the wrong choice for anything else.
PBS (phosphate-buffered saline)
Sodium phosphate plus sodium chloride, buffered to maintain pH near 7.4. Suits assays and cell-culture work that genuinely require physiological pH conditions. No antimicrobial agent. PBS is not a standard injection-grade solvent for research-peptide reconstitution — treat it as the right tool for buffered in-vitro work, not for multi-dose storage.
Why bacteriostatic water is the default
For most lyophilized-peptide reconstitution scenarios, BAC water is the practical default. The case comes down to two things: shelf life and flexibility.
The 28-day rule at 4°C
Reconstituted peptides in bacteriostatic water hold stable for approximately 28 days when refrigerated at 4°C, per widely-referenced USP and vendor guidance. The benzyl alcohol handles the antimicrobial work, preventing bacterial regrowth between draws. The opened BAC water vial itself stays viable for roughly the same 28-day window when refrigerated, so you have two timelines to track in parallel: the BAC vial and the reconstituted peptide vial.
Sterile water has no equivalent guarantee because it lacks a preservative. Reusing a SWFI vial after opening carries a real contamination risk that refrigeration alone does not eliminate.
Long-term freezer storage
At -20°C, peptides reconstituted in bacteriostatic water can remain stable for roughly 3 to 6 months depending on the sequence and storage conditions. Stability beyond that window is better supported for the lyophilized powder form than for reconstituted solutions. If your protocol requires very long storage, keep the peptide dry until shortly before use.
The non-negotiable storage rule applies to every solvent equally: freeze-thaw cycles are destructive regardless of what the peptide is dissolved in. Aliquot before you freeze. The solvent choice is not the fix for freeze-thaw damage; aliquoting is.
When sterile water or PBS is actually the right call
BAC water is not always the right default. Two situations where it is not.
Single-use experiments
If you are reconstituting a peptide for one experiment with no intent to store the solution, SWFI is the cleaner choice. No benzyl alcohol means no preservative interaction with the peptide itself or with a sensitive assay. The trade-off: the reconstituted solution must be used immediately and discarded. Do not store it, even briefly, in an open vial at any temperature.
pH-sensitive peptides and buffered in-vitro work
Some peptides require near-neutral pH to stay in solution and remain functional. PBS at pH 7.4 provides that stability and is appropriate for in-vitro assays, cell viability studies, and receptor-binding experiments where physiological pH is a genuine experimental requirement.
PBS is not ideal where ionic strength or salt interference could skew results, and it is not a standard choice for injection-grade reconstitution. Use it when the experimental design specifically calls for pH buffering — not as a general-purpose alternative to BAC water.
Matching solvent to peptide chemistry
Solvent selection becomes tactical at the amino-acid-composition level. Standard conventions get you started; sequence chemistry gets you to the right answer.
Basic, acidic, and hydrophobic peptides
Basic peptides — rich in lysine, arginine, and histidine — dissolve well in mildly acidic solvents. Dilute acetic acid protonates the side chains and drives aqueous solubility. Standard approach: try water first; if the peptide does not dissolve, acidify before diluting with buffer.
Acidic peptides — rich in aspartate and glutamate — work in the opposite direction. Near-neutral to slightly basic conditions (e.g. 0.1 M ammonium bicarbonate) are the right environment. Pushing an acidic peptide into an acidic solution actively reduces solubility by protonating the carboxylates.
Hydrophobic peptides — more than ~50% non-polar residues — often need an organic co-solvent like DMSO or acetonitrile to break aggregation before dilution with water or buffer.
Cysteine-containing and disulfide-bridged sequences
Peptides containing cysteine or methionine are oxidation-sensitive. Use degassed solvents and avoid combining DMSO with strong acids like TFA, which can drive unwanted oxidation. At pH above 7, single cysteine residues tend to dimerize reversibly, compromising sample integrity. Keeping pH below 7 and working quickly after reconstitution limits this. These solubility requirements should be explicitly addressed in the vendor's product documentation. If they are not, that absence is itself informative — see the next section.
Storage and contamination prevention
Solvent choice is half the equation. Handling decides whether the investment survives the protocol.
Aliquot first, then freeze
The single most important storage habit is aliquoting immediately after reconstitution. Divide the solution into single-use volumes and freeze everything you will not use within 28 days. This eliminates freeze-thaw cycling, which degrades peptide structure cumulatively regardless of solvent choice. Label each aliquot with reconstitution date, solvent used, and concentration. Store at -20°C in small, airtight, sterile vials. Never leave a reconstituted peptide solution in a repeatedly-opened working vial at room temperature between uses.
Aseptic handling
Always use sterile needles and syringes when drawing from a vial. Wipe the septum with 70% isopropyl alcohol before each penetration. For high-sensitivity applications, a laminar-flow hood removes a real variable.
If you see cloudiness, particulates, or unexpected color change in a reconstituted solution, discard it immediately. Those are signs of microbial contamination or peptide degradation that no storage temperature will reverse once they appear.
What vendor reconstitution guidance reveals
The quality of a vendor's reconstitution documentation is one of the more reliable proxies for the quality of what is inside the vial. The connection is worth taking seriously before you place an order.
A vendor that publishes peptide-specific reconstitution guidance — solvent type, concentration range, pH conditions, storage temperatures, oxidation flags for cysteine-containing sequences — demonstrates that they understand the chemistry of what they are selling. Generic instructions like "mix with sterile water," applied uniformly across an entire catalog, are a yellow flag. They often correlate with vendors whose purity scores underperform when independently tested.
This pattern is part of a broader vendor due-diligence framework we cover in detail in 9 Peptide Vendor Red Flags, which lists the nine specific patterns that separate a real research vendor from a wire-fraud trap. Solvent and storage documentation is one of those signals; certificate-of-analysis quality is another. For the broader picture of which compounds are actually backed by the research vendor catalogs claim, see our evidence-based 2026 peptide tier list.
If you want to start with a verified set of vendors instead of working through the entire market, our vendor directory tracks third-party COA history, complaint patterns, and ownership changes across the active US-facing market. Active vendor-quality discussion happens week-over-week in the research forum.
The decision framework, in one paragraph
Use bacteriostatic water as the default for multi-dose research peptide reconstitution — 28-day refrigerated shelf life, multi-month freezer stability with proper aliquoting, contamination protection that single-dose protocols cannot match. Reserve sterile water for injection for clean, single-use experiments where preservative interference is a concern. Reach for PBS when the peptide's amino-acid composition or assay design specifically requires pH stability near 7.4. Then aliquot immediately, avoid freeze-thaw cycles, and apply aseptic technique at every step from solvent draw to vial disposal.
The choice is ultimately inseparable from whether your vendor understands the chemistry well enough to guide you correctly. That is what separates verified, purity-graded suppliers from generic bulk sellers — and it is the question to ask before you mix the next vial.
Frequently asked questions
- What water should I use to mix research peptides?
Use bacteriostatic water (BAC water) as the default for multi-dose protocols — it contains 0.9% benzyl alcohol that prevents bacterial growth between draws and gives you about 28 days of refrigerated stability. Use sterile water for injection (SWFI) only for single-use experiments where you will discard the solution immediately. Use PBS (phosphate-buffered saline) when the assay specifically requires physiological pH near 7.4.
- How long does reconstituted peptide last in bacteriostatic water?
Approximately 28 days at 4°C (refrigerated), per USP and standard vendor guidance. The benzyl alcohol preservative prevents bacterial regrowth each time you pierce the vial septum. At -20°C (frozen), reconstituted peptides typically remain stable for 3–6 months depending on the sequence. Aliquot before freezing — freeze-thaw cycles degrade peptide structure regardless of which solvent you used.
- Can I freeze reconstituted peptides?
Yes, but the right approach is to aliquot first, then freeze. Divide the reconstituted solution into single-use volumes immediately after mixing, then freeze everything you will not use within 28 days at -20°C. This eliminates freeze-thaw cycling, which damages peptide structure cumulatively. Lyophilized (powder) form is more stable than reconstituted solution for long storage — keep peptides in dry form until shortly before use when possible.
- What is bacteriostatic water made of?
Bacteriostatic water is sterile water containing 0.9% benzyl alcohol (9 mg/mL) as a preservative, per USP pharmaceutical specifications. The pH is mildly acidic, sitting around 5.7 with an acceptable range of 4.5 to 7.0. Some peptide-specific BAC water formulations use 0.2% benzyl alcohol to reduce potential interaction with peptide stability — worth confirming on your supplier's product documentation.
- Is sterile water the same as bacteriostatic water?
No. Sterile water for injection (SWFI) is purified, non-pyrogenic water with no additives. It is sterile when sealed but has no antimicrobial protection once opened. Bacteriostatic water adds 0.9% benzyl alcohol as a preservative, which inhibits bacterial regrowth across multiple uses. The practical effect: SWFI is appropriate for single-use only; BAC water can be reused over a roughly 28-day window when refrigerated.